|
Table of Content - Volume 18 Issue 1 - April 2021
Comparison of intravenous tramadol versus intravenous clonidine for control of post spinal anaesthesia shivering
Dhruv Jariwala1*, Malti Pandya2, Divyang Shah3
1IIIrd Year Resident, 2Professor Additional, 3Professor & HOD, Department of Anaesthesiology, SMIMER, Surat, INDIA. Email: dhruvjariwala2012@gmail.com
Abstract Background: Post spinal Anesthesia(SA) shivering an unpleasant event for patient is spontaneous, involuntary, rhythmic, tremor-like muscle hyperactivity that increases metabolic heat production. There are various drugs which have anti-shivering properties. We conducted a study to compare the efficacy of antishivering drug Tramadol and Clonidine for controlling post spinal shivering. Methods: After obtaining well informed written consent, total 60 patients(n=30)were studied when grade 2 to 4 shivering noted for minimum 2 minutes after SA, Inj.Tramadol 1 mg/kg IV(Group-T)or Inj.Clonidine 1 ug/kg IV(Group-C)given. Unpaired t-test and chi-square test were used for data-analysis. Results: Incidence of complete response was more in Group-T wheras Incidence of failure rate, incomplete response and recurrence was less with Group-T.Incidence of PONV were higher in Group-T whereas bradycardia & hypotension were higher in Group-C. Conclusion: IV Tramadol is a better alternative than IV Clonidine in treatment of post SA shivering with prophylactic administration of Ondansetron 4mg IV to prevent PONV. Key Words: Clonidine, post-spinal anaesthesia shivering, tramadol
INTRODUCTION Spinal anaesthesia is widely used as a safe anaesthetic technique for both elective and emergency operations. Post anesthesia shivering is very unpleasant, uncomfortable to the patient as well as to the operating room personnel. It is spontaneous, involuntary, rhythmic, tremor-like muscle hyperactivity that increases metabolic heat production upto 600% after general or regional anesthesia.1 It is a potentially serious complication, resulting in increased metabolic rate; increased oxygen consumption along with raised carbon dioxide (CO2) production; increase in minute ventilation and cardiac output to maintain aerobic metabolism causing arterial hypoxemia, lactic acidosis, increased intraocular and intracranial pressure, and interferes with pulse rate, blood pressure and ECG monitoring by causing artifacts[2].The mechanism of origin of shivering is not clear, various hypothesis have been proposed. Perioperative hypothermia is the primary cause, which occurs due to neuraxial anaesthesia-induced inhibition of thermoregulatory mechanism temperature to maintain internal body temperature within a narrow range, thus optimising normal body function. There are various methods available to control shivering during anaesthesia, which include non-pharmacological methods and pharmacological methods. We conducted this randomised study to compare the relative efficacy of Tramadol and Clonidine for control of intraoperative shivering under spinal anaesthesia.
AIMS AND OBJECTIVES The present study is undertaken to clinically compare the efficacy, haemodynamic effects, complications and side effects of Clonidine and Tramadol on control of postspinal shivering.
MATERIAL AND METHOD After obtaining written and informed consent, we conducted a randomised study in 60 patients and compared the efficacy of Tramadol and Clonidine for controlling postspinal shivering. Patients undergoing various elective surgeries like Hernioplasty, TVH, Tibia nailing, PFN etc. under subarachnoid block who developed shivering after anaesthesia were divided randomly into two groups with 30 patients in each group. Inclusion criteria: Patients from either gender. Aged between 18 to 65 years. ASA grade I or II. Shivering of grade II to IV(CROSSLEY and MAHAJAN) lasting for a minimum period of 2 minutes. Exclusion criteria: Patient refusal; Patients with fever, significant cardiovascular, renal, hepatic, respiratory, thyroid, neurological disorders, autonomic neuropathies, a need for blood transfusion during surgery. Patients with known hypersensitivity to Tramadol or Clonidine; Patients with known history of alcohol and substance abuse; Patients who develop shivering even before administering spinal anaesthesia; Patients requiring supplementation with general anaesthesia; Surgeries which lasted for more than 3 hours Preanaesthetic evaluation Preanaesthetic evaluation of all patients consisted detailed history, physical examination and routine investigation.
OBSERVATION AND RESULTS After studying 60 cases, observation and results were summarized in tabulated form and described below. Both groups comprised of 30 patients.
Table 1: Demographical profile of the patients of both the groups
No significant difference was seen in age, sex, weight, ASA grade, duration of surgery and grade of shivering.
Comparison of axillary temperature(0C) at different time intervals in two study Figure 1: groups The axillary temperature in both groups fall significantly during shivering compared with the baseline values, but the values between two groups did not differ significantly.
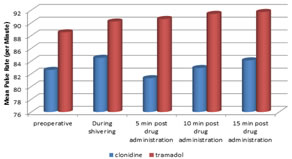
Table 2: Changes in pulse rate (per minute) (mean ± sd)
Comparison of mean pulse rate (per minute) at different time intervals in two study groups
FIGURE 2:
At the onset of shivering, the haemodynamic variables were comparable in both groups. After receiving the study drug treatment, a propensity toward a slight fall in pulse rate was observed in Clonidine group in contrast to Tramadol group, in which no significant haemodynamic changes were observed.
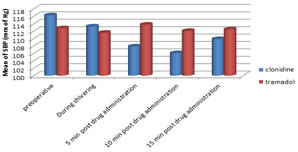
Table 3: Changes in systolic blood pressure (mm of hg ) (mean)
Comparison of mean of SBP (mm of hg) at different time intervals in two study groups FIGURE 3: After receiving the study drug treatment, a propensity towards a slight fall in SBP was observed in Clonidine group in contrast to Tramadol group, in which no significant haemodynamic changes were observed.
Table 4: Comparison of average time taken for cessation of shivering, % of incomplete response, % of no response and % of recurrence in study groups
Time for onset of shivering after spinal anaesthesia was not statistically significantly different between the two groups. The time taken for complete cessation of shivering was significantly higher in Group C than in Group T for which p value was <0.001 indicating that difference is highly significant. The complete response rate was significantly higher for patients treated with Tramadol. The Incomplete response rate, failure rate, recurrence rate was significantly higher for patients treated with Clonidine.
Table 5: Complications in both groups
Incidence of Nausea and vomiting were significantly higher in Tramadol group, whereas Bradycardia and hypotension were higher in clonidine group. Tramadol was found to have less sedation. Episodes of oxygen desaturation or respiratory depression were not detected in any patient of any group during the study.
DISCUSSION Postspinal Shivering is a common problem faced by anaesthesiologist, incidence being 19%-33%. Probable mechanisms could be decrease in core body temperature secondary to sympathetic block; peripheral vasodilatation; increased cutaneous blood flow; which leads to increased heat loss through skin; cold temperature of operation theatre, rapid infusion of cold i.v fluid; and effects of cold anaesthetic drugs upon the thermosensitive receptors in the spinal cord[3,4]. Pharmacological intervention resets the shivering threshold to a lower level, thereby decreasing rigors and its episodes. The neurotransmitter pathways involved in shivering involve opioids, α-2 adrenergic, serotonergic, and anticholinergic receptors.5,6 In present study, we compared the efficacy of Clonidine and Tramadol for post-spinal shivering. Tramadol is a novel analgesic. It has opioid effect mediated by the mu receptor, with minimal effect on kappa and delta receptors. It inhibits 5-HT3 reuptake and promotes its release.It also inhibits synaptosomal noradrenaline reuptake. It also activates the monoaminergic receptors of the descending neauraxial inhibiting pain pathway. The antishivering action of Tramadol is probably mediated via its opioid or serotonergic and noradrenergic activity or both.7 Clonidine is an α2-adrenoceptor agonist. It exerts its anti-shivering effects at three levels: Hypothalamus where it decreases the thermoregulatory threshold for vasoconstriction and shivering, at locus coeruleus -a pro-shivering centre in pons, it reduces spontaneous firing, and at the spinal cord level, it activates the α2-adrenoreceptors and release of dynorphine, norepinephrine and acetylcholine. It is highly lipid-soluble and easily crosses the blood-brain barrier and provides a significant reduction in the incidence of post-extradural shivering without clinically relevant adverse side effects.8,9
INTRAOPERATIVE ENVIRONMENT: Potential risk factors for hypothermia in spinal anaesthesia include aging, level of sensory block, temperature of operation theatre and i.v solutions. Usha Shukla et al., conducted a comparative study of Clonidine and Tramadol for control of post spinal shivering. In either of their study, temperature of i.v fluids, drugs and temperature of operating room were not tightly controlled. However, in our study, the ambient temperature was maintained at 24-26 0C. All preloading fluids and drugs were stored and administered at room temperature. Demographic factors such as age, gender, duration of surgery and anaesthesia have also been matched to reduce any confounding bias. STUDY DRUG AND DOSE: Vyas V. et al., compared efficacy of 1mg/kg Tramadol i.v and 1mcg/kg i.v Clonidine on post spinal shivering. We compared the effect of 1 mcg/kg of Clonidine vs 1mg/kg of Tramadol on postspinal shivering. TEMPERATURE MONITORING: We recorded axillary temperature at regular intervals intraoperatively. PATIENTS INCLUDED, RESPONSE CRITERIA AND RECURRENCE: Usha Shukla et al., included those patients who developed shivering of grade 3 or 4 in their study. In our study, patients with shivering of grade 2 to 4 lasting for min period of 2 minutes were included. Usha Shukla et al., defined response rate as shivering ceasing within 15 minutes after treatment. In our study, shivering control was defined according to grade of shivering after drug administration as either complete, incomplete or failed response. V.Aravind et al., treated recurrence with additional doses of Clonidine 1mcg/kg i.v or Tramadol 1mg/kg i.v in respective groups. In our study, recurrence or incomplete response were treated with active rewarming measures using convection heaters and infusing moderately warmed i.v fluids. We avoided additional doses of study drug and/or multimodal treatment for recurrence so that it does not interfere with intraoperative vital parameters. TIME FOR CESSATION OF SHIVERING: In study by V.Aravind et al., Tramadol took less time (4.58±0.59 min) than Clonidine (8.02±5.15 min) to control shivering. In our study, cessation of shivering with Tramadol was achieved earlier (2.58±0.55 min) than with Clonidine (3.52±0.52 min). Contradictory to this result, Usha Shukla noticed longer time with Tramadol (5.01± 1.02 min) as compared to Clonidine (2.54±0.76 min) for control of shivering. RESPONSE RATE: V.Aravind et al., found Tramadol has significant advantage (100%) over Clonidine (85%) for stopping shivering early i.e at 10 min post shivering. We too found higher incidence of complete response with Tramadol (100%) as compared to Clonidine (80%).In contradiction, Usha Shukla et al. noticed higher success rate in Clonidine group (97.5%) as compared to Tramadol group (92.5%). INCOMPLETE RESPONSE: Pranav Bansal et al. found lower incidence of incomplete response of Tramadol group (26.6%) compared to Clonidine group (46.6%).We too observed incomplete response in 13.3% patients of Clonidine group whereas 0% in patients in Tramadol group. No response was seen in 6.6% patients of Clonidine group. RECURRENCE: Prerna Attal et al., found a higher recurrence rate in Clonidine group (13.3%) than in Tramadol group (6.6%). Our study shows similar findings with higher recurrence in Clonidine group (26.6%) than Tramadol group (6.6%). COMPLICATIONS BRADYCARDIA: Usha Shukla et al., observed incidence of bradycardia was higher in Clonidine group (5%) compared to Tramadol group(0%). Our incidence of bradycardia was also higher in Clonidine group (6.6) than in Tramadol group (0%). HYPOTENSION: Hypotension was seen more frequently in Clonidine group (7.5 %) than Tramadol group (0 %) in Usha Shukla et al. observation. We observed even higher incidence of hypotension with Clonidine (20%) compared to Tramadol (0%) NAUSEA AND VOMITING: Usha Shukla et al. found a higher incidence of nausea (77.5%),vomiting(20%) and dizziness(55.5%) with Tramadol group than Clonidine (0%). Our observation also correlates with above study. Incidence of nausea and vomiting with Tramadol was 50% and 20% respectively which is higher than Clonidine(0%). SEDATION: Prerna Attal et al. observed sedation score of > 2 in more number of patients with Clonidine group (60%) than Tramadol group (20%).In our study, we also found higher incidence of sedation score > 2 in Clonidine group (40%) in contrast to Tramadol group (6.6%).
LIMITATION A limitation of this study is that we could not measure the core body temperature as the probe needs to be put in the oesophagus or near the tympanic membrane. Both these are uncomfortable and unacceptable to the patient who has been given spinal anaesthesia.
CONCLUSION Both Tramadol and Clonidine effectively treated patients with post spinal shivering, but time taken for complete cessation of shivering was earlier in Tramadol than Clonidine; difference being statistically highly significant. Incidence of complete response was more in Tramadol compared to Clonidine. Incidence of failure rate, incomplete response and recurrence was less with Tramadol compared to Clonidine. Incidence of Complications like nausea and vomiting were higher in Tramadol, whereas bradycardia and hypotension were higher in Clonidine. Tramadol was found to have less sedation than Clonidine. From our study we conclude that, i.v Tramadol is a better alternative than i.v Clonidine in treatment of postspinal anesthesia shivering with prophylactic administration of Ondansetron 4mg i.v to prevent nausea and vomiting.
REFERENCES
Policy for Articles with Open Access: Authors who publish with MedPulse International Journal of Pediatrics (Print ISSN: 2579-0897) (Online ISSN: 2636-4662) agree to the following terms: Authors retain copyright and grant the journal right of first publication with the work simultaneously licensed under a Creative Commons Attribution License that allows others to share the work with an acknowledgement of the work's authorship and initial publication in this journal. Authors are permitted and encouraged to post links to their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges, as well as earlier and greater citation of published work.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 Home
Home